Meanwhile, a medical device manufacturer BOSUNG MEDITECH developed blood drawing equipment SafeLan Pro® and all-in-one blood drawing needle, protective cap, and lancet SafeLan® and their function highlighted with user’s safety and hygiene draws attention.
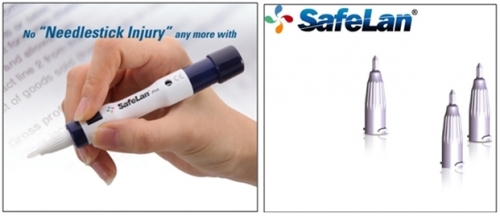
SafeLan Pro® features preventive function for reuse of used lancet. It is designed not to work in any situation unless used blood drawing needle is replaced, and there is a window for loading check to see if blood drawing equipment is ready to use or not preventing user from being pricked by carelessness.
Unlike existing blood drawing equipment which requires complex course of replacing lancet, SafeLan® adopts easy methods of combining and separating and so the protective tip for blood drawing needle is automatically separated only by combining lancet with blood drawing equipment. As blood drawing needle and protective cap are coupled together, the protective cap which leaves bloodstain after drawing blood is discarded with lancet, and it enables prevention of secondary infection caused by bloodstain.
Besides, user’s pain such as ripped skin by trembled blood drawing needle is prevented from inducing for comfortable blood drawing, and as the blood drawing needle is not exposed except the moment of blood-gathering, it can be safely used without being pricked by the needle upon replacing lancet.
SafeLan Pro® and SafeLan®, being approved of such functions, acquired FDA, ISO13485, and CE certifications last November.
BOSUNG MEDITECH’s CEO, Yoon-Chul Shin said “SafeLan Pro® and SafeLan® were developed by focusing on alleviating patient’s pain and operator’s convenience” and “we will develop various disposable sterilized medical devices for the sake of patients and operators”. You can find more information at www.bsmeditech.com and call at +82-33-735-2623
저작권자 © Korea IT Times 무단전재 및 재배포 금지


