SEOUL, KOREA - GE Healthcare announced that it will participate at the Korean Congress of Radiology 2014, which will take place from October 8 to 10 at COEX in Seoul, and introduce innovative medical equipment and solutions that will lead the future of diagnostic imaging.
For the first time, GE Healthcare demonstrates a mock-up Revolution CT (KFDA pending), which was first introduced at ‘RSNA2013’ in December 2013, in Korea. Revolution CT is the first ‘all-in-one’ CT system to overcome the limits of previous CTs in every aspect -- leading to enhanced clinical confidence, fast examination, and wide coverage. The Revolution CT delivers a rotation speed of 0.28s, enabling the production of accurate images even for fast moving organs such as the heart, and the expansion of coverage from 4cm to 16cm helps to produce images of whole organs with excellent image quality. This also enables clinicians to quickly diagnose emergency patients and reduces radiation dose.
Revolution EVO (KFDA pending), one of the Revolution series, is also introduced for the first time at KCR. Revolution EVO features Revolution CT’s Clarity Detector technology that enables clinicians to clearly show details as small as just 0.28 millimeters. It also features ASiR-V, GE’s next generation of model-based iterative reconstruction technology, realizing high-speed, low-dose, and high-quality. ASiR-V reduces dose by up to 82% making it ideal for pediatric scans, oncology and chronic disease follow-up.
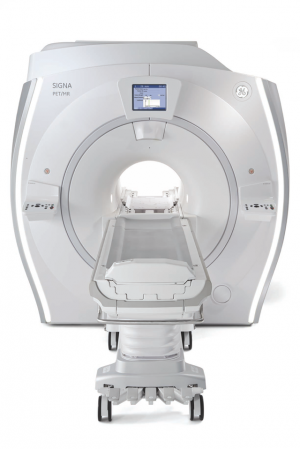
GE’s SIGNATM PET/MR (KFDA pending), which is opening a new chapter in PET/MR, is also introduced for the first time in Korea. PET/MR combines MR and PET scans, enabling clinicians to see early cellular changes that can be accurately mapped onto MR images. With this knowledge, clinicians may be able to shorten the time between diagnosis and treatment offering the convenience of simultaneous PET and MR scans for both clinicians and patients. The 3.0T SIGNATM PET/MR with a 60cm bore features fast coincidence timing resolution enabling Time-of-Flight (TOF) reconstruction and MR Attenuation Correction leading to improved PET image quality with higher structural detail. It also features GE’s exclusive MR-compatible silicon photomultiplier detector (SiPM) technology, which is characterized by its enhanced sensitivity; it is up to three times more sensitive than conventional PET technology. SIGNATM PET/MR is expected to lead to more effective treatment paths for clinicians to offer their patients, particularly for oncology, neurology, and cardiology.
Moreover, GE Healthcare introduces Silent Scan, a revolutionary technology that dramatically quiets MRI exams, with new functions embedded. A new 3D scanning and reconstruction technique called Silenz enables to reduce noise to a level within 3db of the ambient conditions. Now, Silent Scan is also applicable to diffusion-weighted imaging, partial abdominal imaging, and musculoskeletal imaging, which is expected to render MR scanning easier for infants or pediatric patients who were difficult to scan previously.
GE Healthcare’s ultrasound device Logiq E9 dramatically improves diagnostic quality through its ultrasound probe technology XDclear and Shear Wave elastography. XDclear uses a higher frequency by absorbing the heat generated from ultrasound. This enables acquiring extraordinary images even for overweight patients and enhances the resolution of images of the overall abdominal area. With the newly introduced Shear Wave application, LOGIQ E9 uses a focused burst of acoustic energy from the transducer to generate Shear Waves to produce quantitative measurements and a color coded elastogram. These quantifiable exams provide benefits beyond strain elastography, where the operator uses manual compression to produce a qualitative assessment of tissue that can vary from user to user
VolumeRad, a radiographic system, produces as many as 60 high-resolution slice images of the anatomy of interest, including the chest and spine, in approximately 10 seconds. It is 7.5 times more sensitive than chest X-ray in detecting lung nodules and generates clear images at a radiation level below 0.1mSv. Notably, chest exams using VolumeRad are expected to be reimbursed, as VolumeRad has been recognized by the New Health Technology Assessment, a Ministry of Health and Welfare–affiliated organization, as a safe and effective new system.
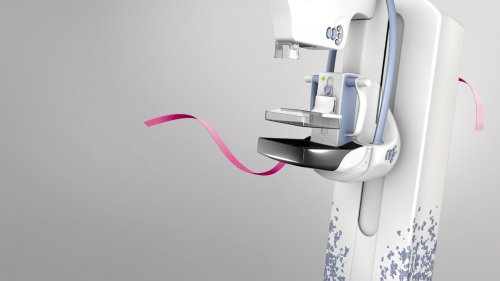
The FDA approved SenoClaire, New Breast Imaging with 3D Tomosynthesis Solution, which has been recently installed in the Seoul National University Hospital, shows 3D image of the breast by using a low-dose short X-ray sweep around the compressed breast with only nine exposures within 10 seconds at a dose level similar to conventional mammograms. This device is expected to substantially enhance clinical confidence in Korean women who have a high rate of dense breasts. It also reduces the pain of patients during the exams as it applies reduced pressure on the breasts compared to conventional mammograms.
Sia Moussavi, president and CEO of GE Healthcare Korea, said, “Korea is respected for its world class radiology society, and we are very proud to be a partner to KCR (Korean Congress of Radiology.” He also added, “Aligning with the theme of 2014 KCR -‘Evidence, Values and Radiology’- we are pleased to introduce our market leading next-generation technologies and solutions to serve Korean radiologists provide improved care with greater confidence in evidence based diagnostics.”
By Lee Jae-seung(jasonlee@koreaittimes.com)


